 
This causes the electrons to experience a greater force of attraction as you move along the period, making it easier for an atoms to gain more electrons to become ions. The charge on the atomic nucleus increases as you move go across the period but the electron shielding caused by the two inner electrons remains the same.The electrons between the elements are shared. Electronegativity is not measured in energy units, but instead a relative scale. The rate at which a chemical substance or an element undergoes a chemical reaction is known as reactivity. 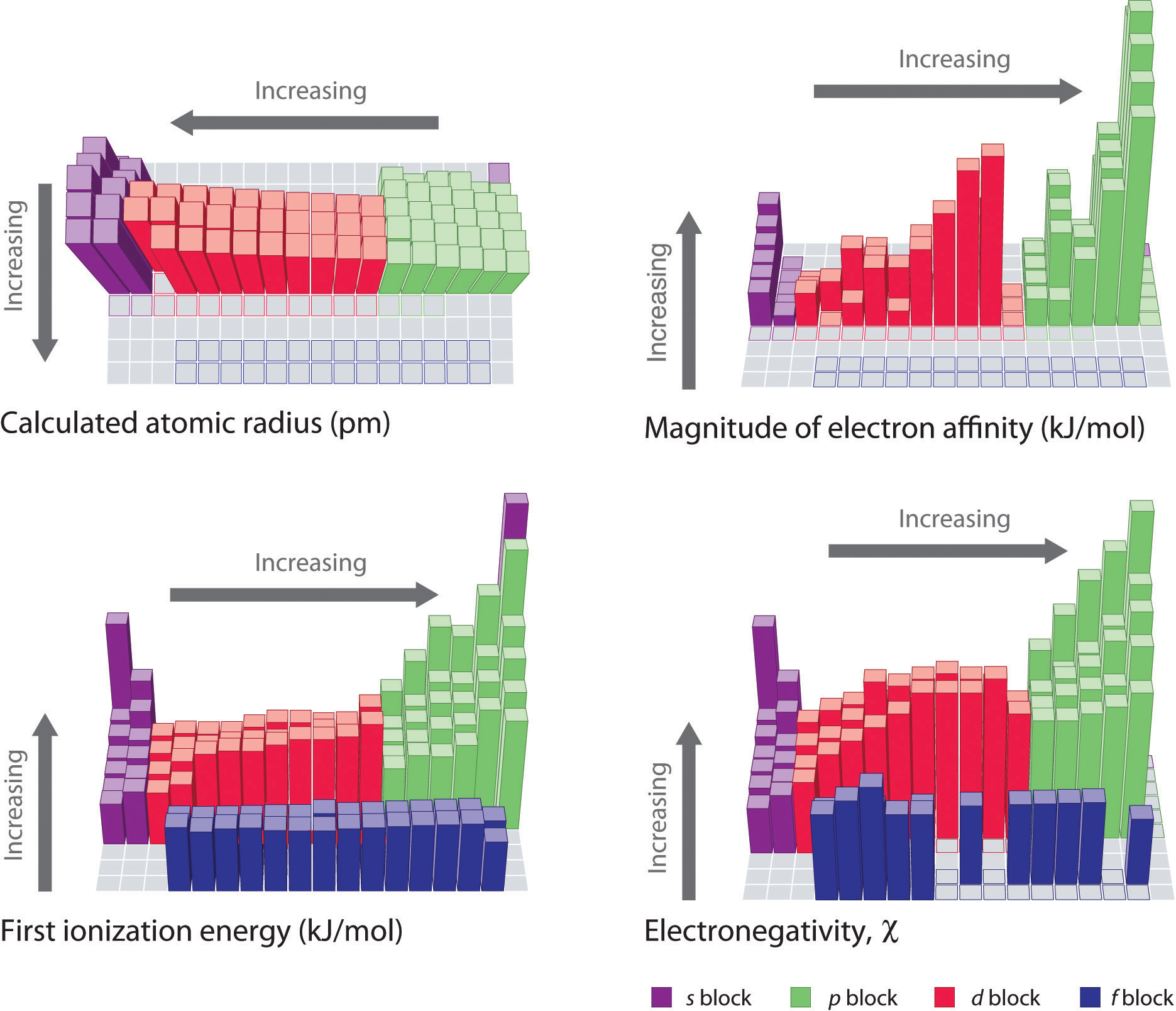
Electronegativity differs from electron affinity because electron affinity is the actual energy released when an atom gains an electron. On the other hand, reactions between nonmetals or between semi-metals and nonmetals invariable form molecular compounds. It is always positive because the removal of an electron always requires that energy be put in (i.e., it is endothermic). Electronegativity is a measure of the ability of an atom to attract the electrons when the atom is part of a compound. The reactivity increases as you go across the period because: Ionization energy (IE) is the amount of energy required to remove an electron from an atom in the gas phase: IE is usually expressed in kJ/mol of atoms. Nitrogen, Oxygen and Fluorine can all gain electrons to become negative ions in certain reactions. Both explanations lead to the same predicted trend. (2) Increasing size of pendant atoms (F < Cl < Br) increases steric repulsions and increases bond angle. This causes the electrons to experience a greater force of attraction as you move along the period, making it harder for the atoms to lose electrons and become ions. The more electronegative pendant atoms can have smaller bond angles. The charge on the atomic nucleus increases as you move go across the period but the electron shielding caused by the two inner electrons remains the same. Moving left to right across a period, atomic radius decreases, so electrons are more attracted to the (closer) nucleus.It uses these reactions to explore the trend in reactivity in Group 1. Explaining the reactivity trend of the Group 7 halogens. OxidationReduction theory of halogen displacement reactions. The reactivity order and halogen displacement reactions. Covalent and ionic bonding in Group 7 halogen compounds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
